Energy forms and conversions
Energy is a fundamental quantity in nature that is transferred between systems or parts of systems. It is ‘stored’ in a variety of forms and is associated with movement, combustion, heat and (the capacity to do) work. Boiling water for a cup of tea, using the microwave oven, switching on the central heating system, cooling food in the refrigerator, lighting a room, riding a bicycle or driving a car are all processes in which one form of energy is converted into another. The conversion processes are described in scientific equations and models that have evolved during centuries of experimentation. This introduction touches only upon a few aspects of heat and mass transfer processes of relevance for the notion of energy and the transition to a sustainable energy system.
Energy is usually estimated for the various end-uses in units of (primary or secondary) energy carriers. However, what physically matters is the form of the energy, one important distinction being between stored energy and transitional energy. Stored energy is energy that is available in a mass or a position in a force field (gravitational, electromagnetic, chemical). Stored energy can be converted to another form of stored energy and the process takes place via transitional energy: heat Q and work W.
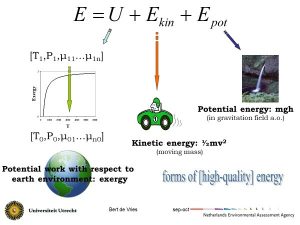
Two forms of energy are potential and kinetic energy in a gravitational or electromagnetic field. A lifted arm or object represent potential energy in the earth gravitational field. Water flowing downhill, a moving bus or a metal rod spinning in a fluctuating magnetic field in an operating electromotor represent kinetic energy. Another form is the energy of the constituent atoms and molecules in the form of rotational, vibrational, translational and chemical bonding energy of atoms and molecules. It is denoted as internal energy U. It is contained in chemical and nuclear bonding and is manifested as sensible heat (temperature related) or latent heat (phase change related, for instance, when water vapour condenses as raindrops). Changes in potential, kinetic and internal energy is what matters in human use. Table 1 is a matrix of the various energy conversions and the processes and appliances in which they take place.
Since the 19th century, two laws of thermodynamics have been formulated and reformulated. In essence, they state that for an isolated system there is a quantity called energy which is constant (‘conservation of energy’) and that there is another quantity called entropy which can never decrease and tends towards a maximum in an equilibrium state (‘arrow of time’). It expresses the natural tendency towards disorder One consequence is that work W can always be converted to heat Q but not vice versa. Potential and kinetic energy can in principle entirely be converted into work. The other way around, the internal energy U once manifesting as heat can only partly be converted into work. Hence, work is the high-quality energy form and of most interest for human use. The essential energy flow on earth is solar electromagnetic radiation, which is converted into heat upon insolation and to chemically stored energy via photosynthesis in plants. The plants can under geological forces turn into stores of chemical energy (‘fossil fuels’) and can be transformed in the human or animal body into heat and work kinetic (metabolism). Solar radiation can partly be converted directly into electricity, in photovoltaic (PV) solar cells, or into heat, in solar heat absorption collectors. The chemical energy can partly be converted into electric power in a steam or combustion engine or turbine. chemical gravita- tional electric electro- magnetic nuclear kinetic/mechanical displacement elevator generator magnetic nescence (laser display) mechanical hydroturbine Table 1. Energy forms and conversions. Each column shows how a particular form of energy can be converted into another one that is indicated in the corresponding row to the left. Many of the laws and models describing real-world near- or non-equilibrium processes are empirical or phenomenological laws. The process of heat transfer is elementary and provides direct insights into ‘energy savings’. Heating air in a room with a stove or mass in a furnace is a heat transfer process. No work is involved: you simply produce a flow of heat Q from burning a fuel. As soon as the air or the mass has a temperature higher than the ambient temperature, Nature strives to equilibrate the two – the consequence of the Second Law. The heat transfer through conduction from the room with an average air temperature Troom to the surroundings where the air has an ambient temperature T0, is approximated by Fourier’s Law: The heat flow Q in Joule out of the room in a time period of t in sec is proportional to the thermal or heat conductivity k (in J/m/oC) of the material and area A (in m2) of the interface (wall, window) between room and surroundings and to the temperature difference (Troom – T0) or temperature gradient in oC between inside and outside across the thickness of the interface Δx (in m) [4]. This relationship is essential knowledge if you want to insulate your house or an engineer wants to reduce process heat losses. Thus, you can reduce the heat flow (and the energy bill) by: The third option, known as energy conservation or ‘energy saving’, is now widely applied and new buildings have to comply with ever stricter standards [6]. With t = 1 second, the proximate k-values are in 0,2-2 W/m/K for concrete, 0,8-0,9 W/m/K for glass, 0,5-0,7 W/m/K for bricks, 0,1-0,5 W/m/K for wood, 0,05 W/m/K for mineral wool and 0,025 W/m/K for polyurethane (PUR). Insulation is very effective: replacing a single-pane window with the latest HR-glass lowers k from 6 to 1 W/m2/K – or 1 kWh per m2 of window on a cold day. Also, it reduces noise levels. Putting glass mineral wool under the floor or against a standard roof gives similar reductions. Innovations lead to ever-lower heat conductivity values, but it takes time before they are cost-competitive and accepted by house builders and owners. Two other processes for which universal the descriptions have emerged are diffusion and discharge of mass flows. Mass flows, and their diffusion in air, water and soils are ubiquitous in biological and environmental as well as in technical processes. They are the core of micro-level understanding of processes such as changes in soil nutrients, dispersion of pollutants and (bio)accumulation. There are two empirical laws that formulate the change in terms of a gradient. The first one is Fick’s (first) law about the diffusion of a substance across a concentration gradient. In equation form: with m the mass, A the diffusion surface, (ch – cl) the concentration gradient between inside and outside across the thickness of the interface Δx (in m). D is the diffusivity which is the equivalent of conductivity in Fourier’s law. If concentration is replaced by osmotic pressure, it describes the process in which osmotic pressure difference is used to extract work from the difference in salt concentration at the outflow of river into the sea. A Norwegian pilot plant uses this salinity gradient to generate 2–4 kW of power, at 1 W/m2 and flow rates of 10–20 litre/s. It is a truly renewable resource, but the membranes are still very expensive. A similar relationship is used to describe the discharge of a fluid in the soil. For instance, water flows in an aquifer are determined by the pressure drop per unit length and are approximated with Darcy’s law: with V the volume, k the permeability, A the discharge surface, μ the viscosity and (Ph – Pl) the pressure gradient between inside and outside across the thickness of the interface Δx (in m). Here, the inverse of viscosity is the equivalent of conductivity. It is the basic equation for the calculation of the energy needed to pump up water – or oil or gas – from an underground reservoir. These laws, and other ones like Ohm’s law for electrical current and the Gaussian dispersion model for air pollutants, express the second law observation that Nature tries to reduce the difference in intensive variables (temperature, pressure, concentration) between two (parts of) systems. The equilibrating process is a negative feedback loop (§9.2). These laws are never exact in the real world, their relevance is in describing the underlying process and being often a good first approximation or ‘rule-of-thumb’ (§8.6). Interestingly, these laws came into existence well before they were scientifically understood at the micro-level and were based on the intuition that inhomogeneities spontaneously tend to dissolve at a rate proportional to the gradient, that is, on the Second Law. Figure 1 Three forms in which exergy is available Figure 2 Three forms of energy: potential energy, kinetic energy and (work from) heat Classical thermodynamics is, in a sense, a strange science. It can only make statements about systems in equilibrium, that is, systems in which no change and no outside intervention takes place [7]. In equilibrium, the state variables have fixed values and thermodynamics can make statements about processes from an equilibrium state 1 to an equilibrium state 2. The change from state 1 to state 2 is caused by interaction between the system and its environment, for instance by adding an amount of heat Q to a cylinder of air while fixing the volume V or by extracting an amount of work W from it by letting it expand. There are in principle infinitely many ways to bring the system from state 1 at time t to state 2 at time. What tells thermodynamics about such processes? Energy forms are not equivalent. Whereas the First Law of thermodynamics states the conservation of energy, the Second Law introduces quality of energy in the form of the concept of entropy. Every real-world conversion process will degrade the quality of the energy in dissipative processes such as friction. Macroscopic order, in the form of, for instance, potential or kinetic energy, is converted into microscopic disorder in the form of heat.The concept of entropy S is defined in such a way that increasing disorder in a system is equivalent to an increase in entropy. Processes of heat transfer, mass diffusion and others are driven by the gradients in the intensive variables such as temperature, pressure or concentrations. The larger the gradient, the larger the driving force, the more irreversible the process – and the larger the dissipative work. If you drive a distance of 110 km in a car in two hours at 55 km/hr (~15 m/s), you need about 5 kWh to overcome air resistance. If you drive the same distance in one hour at 110 km/hr (~30 m/s), you need 20 kWh or four times more (MacKay 2009). But large driving forces are desirable because they make processes go fast, and we humans are in a hurry because we do not live forever. Doing things means loss of potential work – and doing things fast means more loss of potential work. Or the other way around, doing things slowly reduces loss of potential work. Of course, doing things infinitely slowly makes no sense, so there is some optimum between what we want to accomplish and how much work potential we are able and willing to use for it. The second law of thermodynamics states that entropy is produced whenever the change process is not infinitely slow. This stems from the different forms of friction if the process proceeds at finite velocity – think of the turbulence that you create stirring your cup of coffee. One consequences is that only a fraction (1 − Tl/Th) can be extracted in the form of work from an energy flow between a heat source at temperature Th and a heat sink of temperature Tl (Figure 1). To extract this maximum amount of work, the process has to happen infinitely slow and reversibly [8]. It confirms the intuition that heat has a lower quality than other forms of energy: only part of it can be converted into work! In qualitative terms, it expresses the fact that heat is a less ordered form of energy than potential and kinetic energy in force fields. The concept of energy quality can now be operationalized by the realization that the earth is the ultimate environmental sink, at ambient temperature T0. In other words: the temperature gradient tells how much work can maximally be extracted from the system by bringing it in equilibrium with the ambient temperature [9]. In the real world, the work W that can be extracted in a change from state 1 to state 2 will always be less than the fraction (1 – T0/Th) because of the irreversible entropy production. This maximum amount of work that can be extracted from a given amount of energy against a well-defined earth environment is the exergy ɸ of the system. Unlike energy, exergy is ‘consumed’ and lost during a conversion process. Because work is what we want, exergy provides a ranking of energy stocks and forms according to quality. Table 2 lists the quality of energy and matter in a system in terms of the exergy content. Of particular interest is combustion (‘burning’) of carbon fuels in which chemically stored energy in the reactants (‘fuel’ and air) is released in the form of heat in the products (mostly water and carbon dioxide) of temperature T. The higher the temperature of the combustion products, the more work can be extracted (Figure 3). The exergy of fuels is for most fuels within 5% of the combustion enthalpy. The sun can be considered a black body of 6000 (degree Kelvin) K according to Planck’s law. Therefore, solar radiation has in theory about 95% work potential and is a high-quality energy source. Nuclear energy in fission material and chemical energy in fossil fuels or biomass can be converted into a heat flux of high temperature – usually in the range of 500◦C to 1500◦C – and, therefore, they represent stored high-quality energy. Table 2 The quality of different forms of energy. The quality index indicates the fraction (in %) of an amount of energy that can be converted to work by bringing it reversibly into equilibrium with earth environment (after Wall 2001; www.exergy.se). Why is the notion of energy quality important? Heat is not the most interesting form of energy: you probably intuitively grasp that one kWh of heat in your bathtub is less or at least differently useful than one kWh from an electrical outlet in order to power your laptop or charge your battery. Although human beings have always appreciated heat as a way to make their habitat more accessible and comfortable and to perform tasks like cooking and metal smelting, it really is work that matters most. Farming requires ploughing, irrigating, terrace building – traditionally done by human and animal muscle power, and in some places by wind or water. Manufacturing requires moving around materials and products, compressing, cutting, separating and so on – processes which could only be done at large scales and rates after the introduction of the fossil-fuel based steam engine and later steam or gas turbine. Transport means moving people and goods around – traditionally done with animals but nowadays almost completely based on the internal combustion engine. Finally, communication increasingly means moving electrons around. Literature Blok, K., and E. Nieuwlaar (2020). Introduction to Energy Analysis. Routledge MacKay, D. (2009). Sustainable Energy- without the Hot Air. UIT Cambridge UK Footnotes [1] How then is the order that we see in organisms, humans or societies possible? The answer is that the emergence and sustenance of complex highly-ordered systems is only possible at the expense of an increase of disorder in the environment. Were the earth an isolated system, life would be (nearly) impossible. But the earth is not: the solar energy influx represents an energy source from which work can be extracted before it dissipates into ambient-temperature heat – in photosynthesis and in photovoltaic cells, for instance. [2] In an electrochemical or fuel cell, the conversion rate to electricity has in theory no inherent limitation. But there is still a gap between theory and practice, and fuel cells are not yet a reliable and cheap enough option for large-scale application. [3] Around 1900, power plants worked with low exhaust temperatures and had efficiencies of 5–10%. Modern power plants are operated in a water-vapour Rankine-cycle steam or in a gas turbine or a combination of both: a so-called Steam And Gas Generating-Combined Cycle (STAG-CC) plant. Advances in mechanical and materials science and technology make it possible to operate STAG-CC plants with inlet temperature of 1200K or more, reaching exergetic (chemical-to-electric) efficiencies up to 60%. One can also increase the energetic efficiency by using the low-temperature heat from exhaust gases to heat offices or dwellings (district heating) or for use in industrial processes (Combined Heat and Power or CHP). [4] The temperature (difference ) is usually indicated in degree K(elvin). [5] An alternative formulation is in terms of the heat resistance R = d/k in m2K/W, with d the thickness of the layer. [6] See the website https://en.wikipedia.org/wiki/Thermal_conductivity for a list of k-values. [7] This shows the huge simplification of economic processes when they are described in the conceptual framework of classical thermodynamics, as is done in mainstream economic theory. [8] For a more in-depth analysis, I refer to standard textbooks on thermodynamics. [9] Pressure and concentrations are other intensive variables with which a system can be brought in equilibrium. Therefore, when a system deviates from ambient pressure p0 and concentrations μi0, it has a work potential due to the gradient (p-p0) and (μi– μi0).
FROM:
thermal
TO:
thermal
heat flow (heat exchanger), phase transition (boiling, melting etc.)
exothermal reaction, combustion
resistance heating
solar heat collector
fission/fusion (nuclear reactor)
friction in movement, inelastic collision
chemical
endothermal reaction, thermolysis
chemical reaction
electrolysis (battery charging)
photo-synthesis
ionisation
radiolysis
gravita-tional
mass
electric
lifting a mass
electric
thermo-electricity
battery, fuel cell
transformator
solar (PV) cell
röntgen
dynamo
electro-
thermal radiation (light bulb), thermocouple MHD-generator
chemolumi-nescence fire flies
electrolumi nescence (LED-lamp), radio/tv, LCD-display etc.
photolumi-
radioactivity
axe on stone (sparks)
nuclear
particle accelerator
gamma reactions
charged particle reactions
kinetic/
expansion, heat engine, (internal combust, steam/gas turbine)
metabolism, muscle power, fire arms
falling mass
electric motor, flywheel
solar sail
particle emission (nuclear bomb)
gearbox
 The most important conversion is from heat to electric power. Physically speaking, electric power is a flow of electrons in a conductor. By analogy with water, one can speak of pressure or force (Volt), flow (Ampère) and power equal to pressure times flow (Watt) in a pipe (conductor). The pressure or force to move the electrons has to come from movement in a magnetic field, which is the core process in electric power generators and dynamos. The movement can come from water or wind turbines or from steam of gas turbines driven by heat from fuel combustion, geothermal sources or nuclear fission. If the heat flow comes from fuel combustion, the chemical energy in the fuel can in theory be converted into electricity with close to 100% exergetic efficiency [2]. The higher the exhaust gas temperature expanded in a turbine and the lower the temperature of the at ambient river or sea water (condenser) or air (cooling tower) to which heat is exported, the lower the exergy losses [3]. The other option to generate shaft work for generating electric power are the internal combustion (Otto and diesel) engines and the Stirling engines. They are much smaller, they are used primarily in cars and ships and for cogeneration and their efficiencies are (much) lower.
The most important conversion is from heat to electric power. Physically speaking, electric power is a flow of electrons in a conductor. By analogy with water, one can speak of pressure or force (Volt), flow (Ampère) and power equal to pressure times flow (Watt) in a pipe (conductor). The pressure or force to move the electrons has to come from movement in a magnetic field, which is the core process in electric power generators and dynamos. The movement can come from water or wind turbines or from steam of gas turbines driven by heat from fuel combustion, geothermal sources or nuclear fission. If the heat flow comes from fuel combustion, the chemical energy in the fuel can in theory be converted into electricity with close to 100% exergetic efficiency [2]. The higher the exhaust gas temperature expanded in a turbine and the lower the temperature of the at ambient river or sea water (condenser) or air (cooling tower) to which heat is exported, the lower the exergy losses [3]. The other option to generate shaft work for generating electric power are the internal combustion (Otto and diesel) engines and the Stirling engines. They are much smaller, they are used primarily in cars and ships and for cogeneration and their efficiencies are (much) lower.Elementary energy conversion processes
Energy quality: exergy
Form of energy/matter
Quality index (%)
Extra superior
Potential energy, e.g., high water reservoir
100
Kinetic energy, e.g., waterfalls
100
Electrical energy
100
Pure element, e.g., diamond
100
Superior
Nuclear energy in nuclear fuel
almost 100
Metals (commercial quality)
almost 100
Sunlight
95
Chemical energy in fossil fuels
95
Element mixes, e.g., steel, alloys, plastics
90
Rich mineral ore deposits
50–80
Hot steam
60
Poor mineral ore deposits, e.g., bauxite
20–50
District heating
30
Water in radiator at 50°C
15
Inferior
Waste heat
5
Valueless
Heat radiation from the earth
0


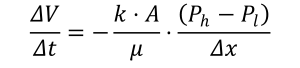

Leave A Comment